 Also note the first peptide chain possesses an internal loop. He found the primary structure to comprise of two chains linked by two cysteine disulfide bridges. This pioneering work, completed in 1953 after some 10 years of effort, earned a Nobel Prize for British biochemist Frederick Sanger (born 1918). Insulin was the first protein whose amino acid sequence was determined. Remember that reduction is the addition of hydrogen.Ĭysteine residues in the the peptide chain can form a loop buy forming the disulfide bond (-S-S-), while cysteine residues in different peptide chains can actually link what were otherwise separate chains. The reduction of a disulfide bond is the opposite reaction which again leads to two separate cysteine molecules. (The insulin molecule shown here is cow insulin, although its structure is similar to that of human insulin. For example, the hormone insulin has two polypeptide chains, A and B, shown in diagram below. This is an important bond to recognize in protein tertiary structure. The backbone of a protein is composed of amide bonds, which are well solvated by (complexed with) the surrounding water. The simplest level of protein structure, primary structure, is simply the sequence of amino acids in a polypeptide chain. The sulfurs (yellow) join to make the disulfide bridge. An unspecified oxidizing agent (O) provides an oxygen which reacts with the hydrogen (red) on the -SH group to form water. The oxidation of two cysteine amino acids is shown in the graphic. The oxidation of two sulfhydryl groups results in the formation of a disulfide bond by the removal of two hydrogens. The amino acid cysteine undergoes oxidation and reduction reactions involving the -SH (sulfhydryl group). An important resonance contributor has a C=N double bond and a C-O single bond, with a separation of charge between the oxygen and the nitrogen.Īlthough B is a minor contributor due to the separation of charges, it is still very relevant in terms of peptide and protein structure – our proteins would simply not fold up properly if there was free rotation about the peptide C-N bond.ĭisulfide Bridges and Oxidation-Reduction This, along with the observation that the bonding around the peptide nitrogen has trigonal planar geometry, strongly suggests that the nitrogen is sp 2-hybridized. One of the most important examples of amide groups in nature is the ‘peptide bond’ that links amino acids to form polypeptides and proteins.Ĭritical to the structure of proteins is the fact that, although it is conventionally drawn as a single bond, the C-N bond in a peptide linkage has a significant barrier to rotation, almost as if it were a double bond. This results in a polypeptide chain that twists in a regular coil shape with the R-groups pointing outwards away from the peptide backbone. 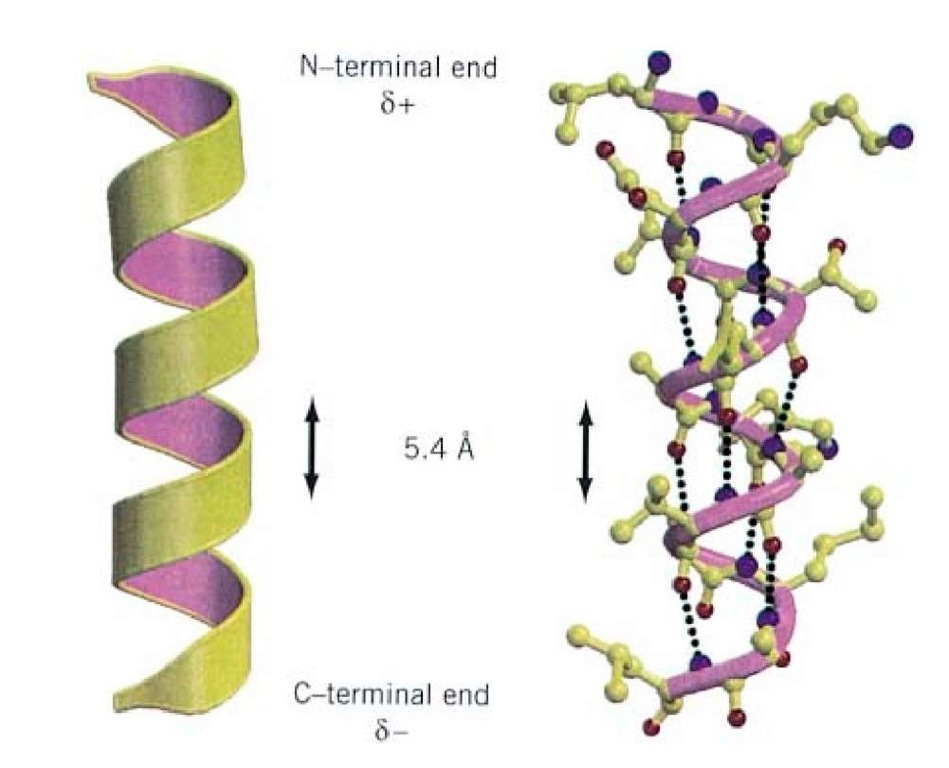
Resonance contributors for the peptide bondsĪ consideration of resonance contributors is crucial to any discussion of the amide functional group. The -helix is a right-handed coil in which backbone NH group hydrogen bonds to the backbone C O group of the amino acid located four residues earlier along the protein sequence. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
